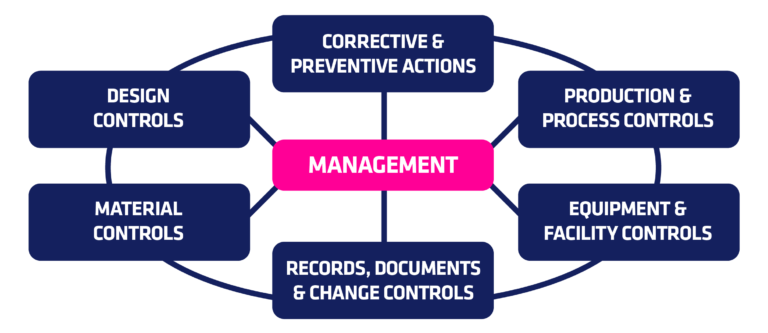
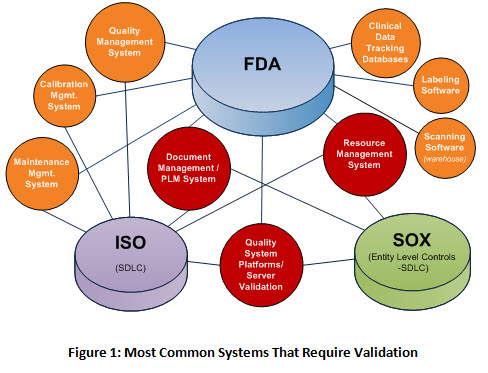
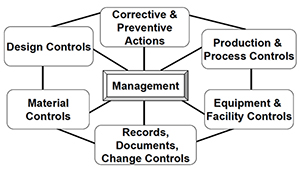
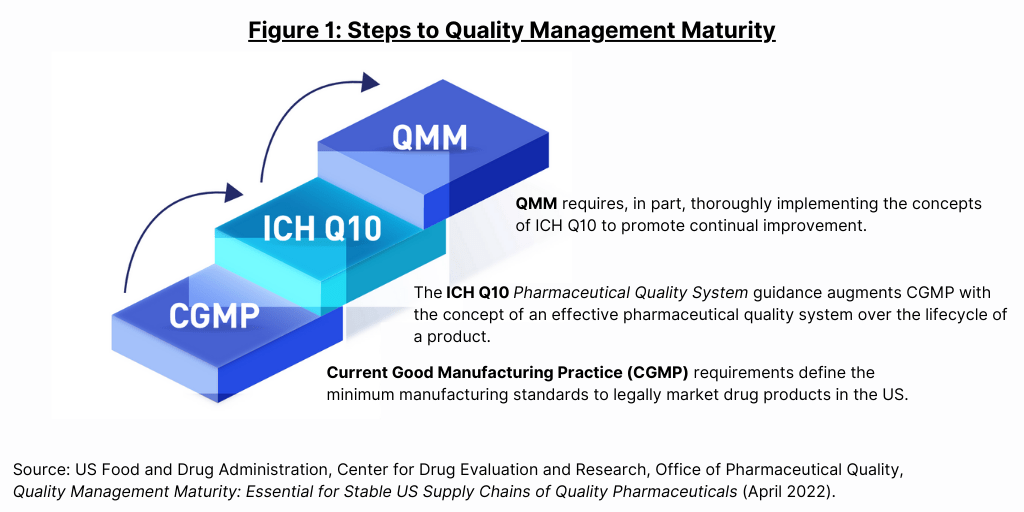
Live from PDA/FDA: FDA Considers Incentivizing Quality Management Maturity (QMM) | Healthcare Packaging

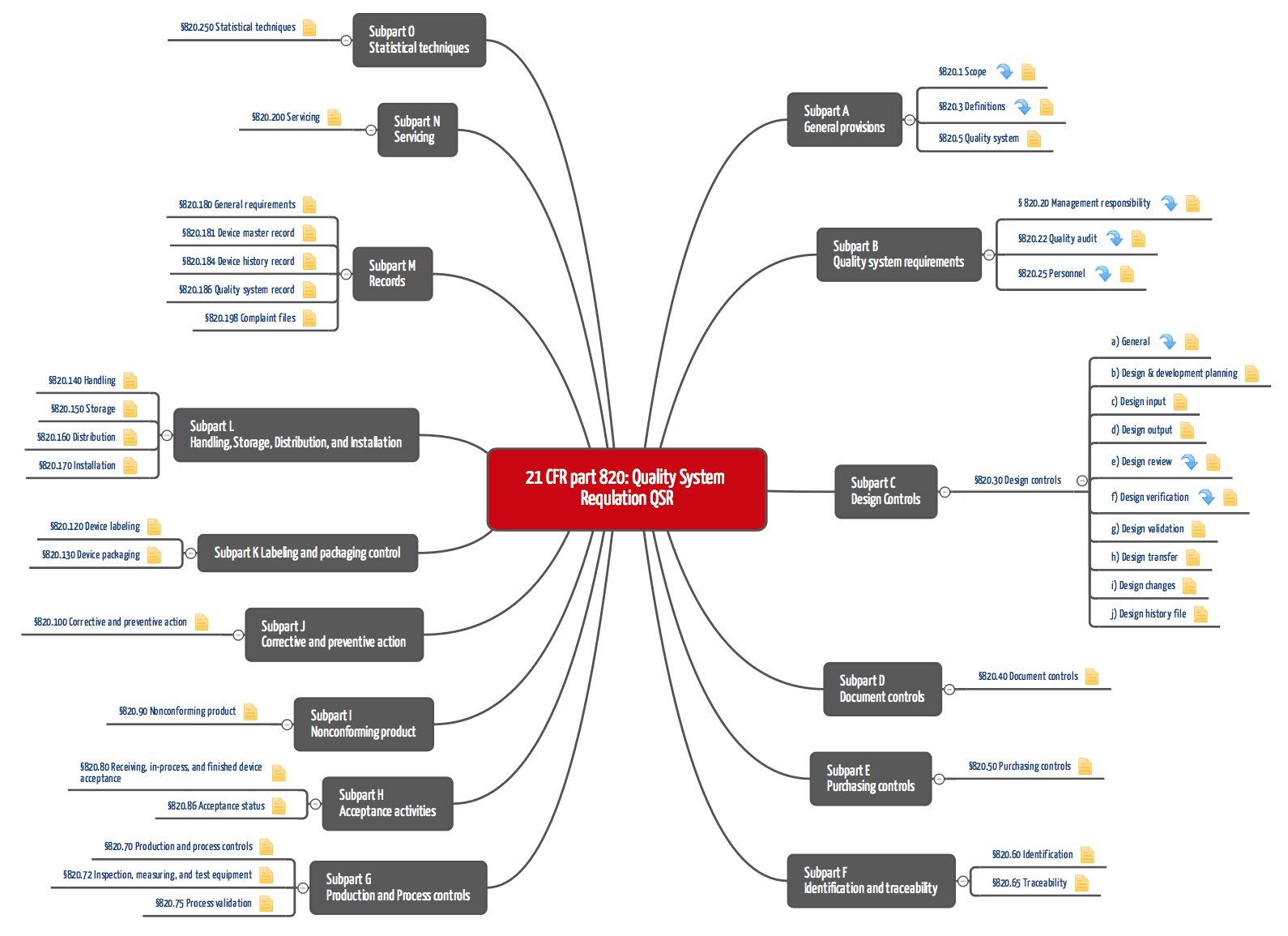
Designing A World-Class Quality Management System For FDA Regulated Industries: Quality System Requirements (QSR) For cGMP : Muchemu, David: Amazon.fr: Livres

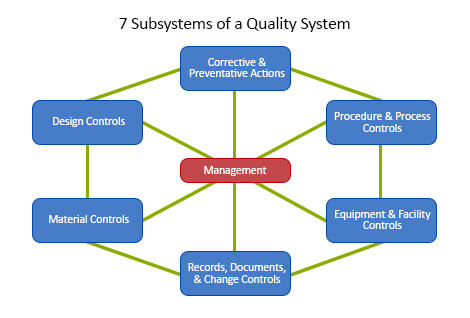
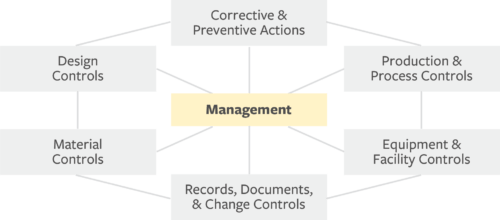
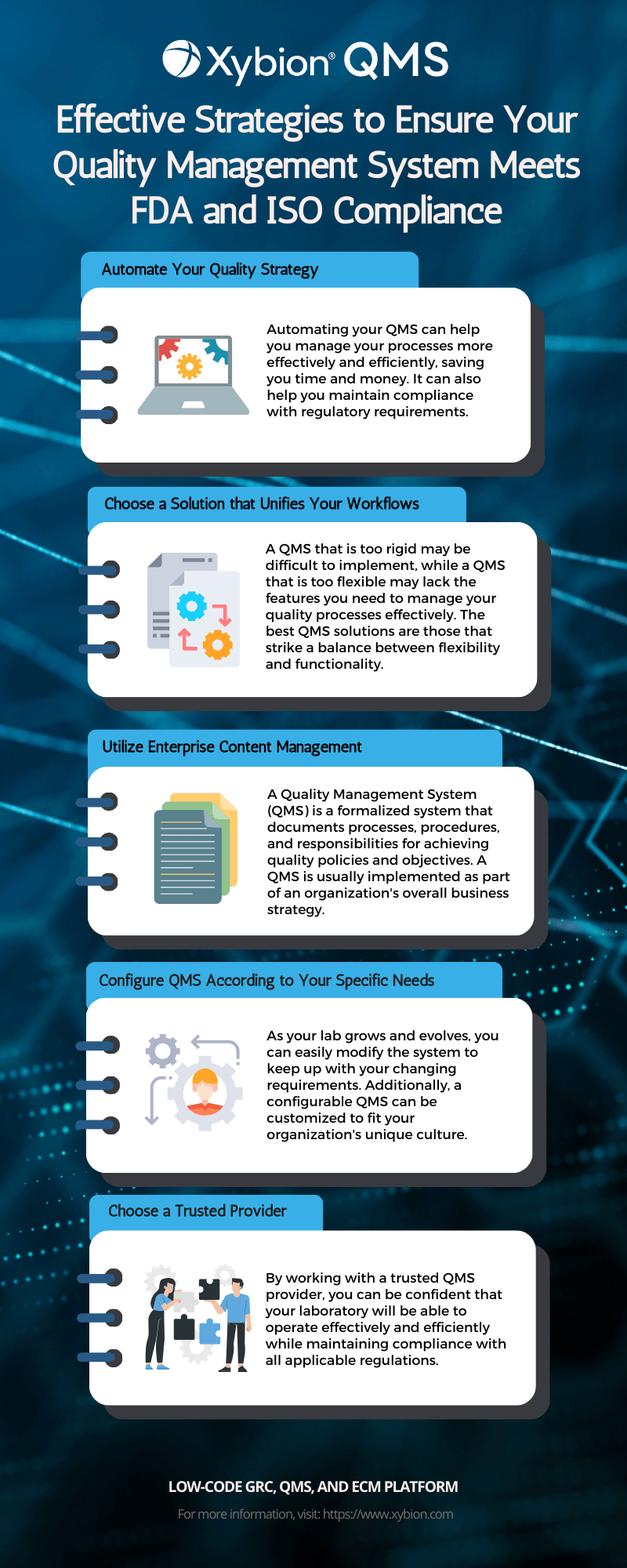
.webp?width=500&height=381&name=Quality%20management%20system%20product%20development%20(1).webp)